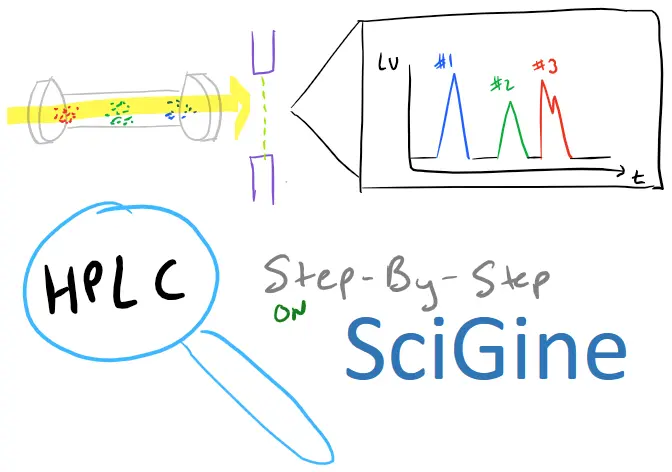
Hewlett Packard HPLC 1090 is a high-performance liquid chromatography instrument that is widely used in analytical laboratories for the separation and analysis of chemical compounds. In this article, we will provide a step-by-step guide on how to start and operate the HPLC instrument, as well as discuss the different types of chromatography and their applications.
How do I start my HPLC instrument?
Starting an HPLC instrument requires careful preparation and setup. Here is a complete step-by-step guide:
Materials
- HPLC autosampler vials
- Centrifugal filters with 0.2 um pores
- Eppendorf vials
- HPLC machine
Methods
In a typical HPLC procedure, you can decide the following variables:
- Flow rate: Determines the speed at which peaks come out. Higher flow rates result in faster elution but may lead to peak blending.
- Pressure: Affected by the flow rate and solvent. It is important to ensure stable pressure during the analysis.
- Solvent Buffers: Determines signal intensity, peak elution time, and signal fidelity.
- Column Type: Determines the type of interaction with the sample. Common types include normal phase and reverse phase chromatography.
- Detection Parameters: If using UV or fluorescence detectors, you need to set the appropriate excitation and emission wavelengths.
For sample preparation:
- Dissolve your biopolymers or small molecules in a suitable solvent such as methanol.
- Centrifuge the sample at 10,000 rcf in an Eppendorf vial to remove any large particulate matter.
- Use a centrifugal filter to further clean up the sample by adding 500 ul of the sample solution and centrifuging at 10,000 rcf.
- Load the filtered sample into an HPLC vial.
For setting up the HPLC machine:
 Revolutionizing it infrastructure: hp hyper converged solutions
Revolutionizing it infrastructure: hp hyper converged solutions- Make sure you have all your buffers prepared and ready.
- Open the purge valve and purge the system for 5 minutes to remove any air bubbles.
- Add your samples into the autosampler tray.
- Close the purge valve to stop the purge.
- Run the system at a normal flow rate (1 ml/min) with your buffer to equilibrate the column for 10 minutes.
- Ensure that the pressure is stable, with minimal fluctuation.
- Set up your sequence and method for the analysis.
- Run a standard before your actual samples to calibrate the instrument.
Here's an example buffer system to determine Fluoresceinamine levels in samples:
Sample: Add 10 ug of fluoresceinamine into 1 ml of Acetonitrile.
Buffer: Pure acetonitrile buffer on a C-18 column; this is a reverse phase chromatography.
Flow rate: 1 ml/min.
Column: 6 mm x 30 cm size.
 Troubleshooting hp photosmart 6520: black ink not printing
Troubleshooting hp photosmart 6520: black ink not printingDetection: Detect via a fluorescence detector set to Excitation @ 485 nm and Emission @ 535 nm.
Notes on HPLC methodology:
- To clean the system and equilibrate it, you need to run enough solvent. However, the amount varies depending on the column size.
- Isocratic means that the solvent concentration stays constant throughout the run.
- Running standards before and with your samples helps in identifying the peaks of interest.
- Always use HPLC grade solvents to ensure accurate results.
Types of Chromatography
HPLC utilizes different types of chromatography to separate and analyze chemical compounds. The two main types are:
Normal Phase Chromatography
In normal phase chromatography, the column is filled with polar silica particles, and the buffer running through the system is non-polar. This results in polar molecules sticking to the silica more and having a longer retention time than non-polar molecules.
Reverse Phase Chromatography
In reverse phase chromatography, the column is filled with hydrophobic particles, usually silica particles with long hydrocarbon chains on the surface. The buffer running through the system is polar, such as acetonitrile/water or methanol/water mixtures. This causes hydrophobic molecules to stick to the resin more and be retained longer.
 Hp40gs graphing calculator: the ultimate tool for advanced math
Hp40gs graphing calculator: the ultimate tool for advanced mathApplications of HPLC
HPLC has a wide range of applications in various fields, including pharmaceuticals, environmental analysis, food and beverage industry, forensic science, and more. It is used for the analysis of biopolymers, small molecules, and polymers. Some common applications of HPLC include:
- Determination of drug impurities
- Analysis of amino acids and peptides
- Quantification of pesticides in food samples
- Identification of environmental pollutants
- Quality control analysis in pharmaceutical manufacturing
In conclusion, Hewlett Packard HPLC 1090 is a powerful instrument used for high-performance liquid chromatography analysis. By following the proper setup and method guidelines, you can effectively separate and analyze chemical compounds for various applications. Whether you are a researcher, scientist, or laboratory technician, HPLC is an essential tool in your analytical arsenal.
References
- ChemGuide Summary of Technique - [link to the source]
- Method Guide from Waters - [link to the source]
- Overview on Wiki - [link to the source]
Q: What is the purpose of HPLC?
A: HPLC is used for the separation and analysis of chemical compounds, including biopolymers, small molecules, and polymers. It provides valuable information about the composition and properties of these compounds.
Q: What are the advantages of using HPLC?
A: HPLC offers high resolution, sensitivity, and versatility in separating complex mixtures. It allows for the quantification and identification of compounds in various samples.
Q: Can HPLC be used for quantitative analysis?
A: Yes, HPLC is commonly used for quantitative analysis due to its accuracy and precision in measuring compound concentrations.
 Exploring hewlett packard & htc: innovations & flagship products
Exploring hewlett packard & htc: innovations & flagship productsQ: How does HPLC differ from other chromatography techniques?
A: HPLC utilizes a high-pressure liquid mobile phase to separate compounds, whereas other chromatography techniques may use different mobile phases such as gas or thin-layer chromatography.
Q: Can I use HPLC for analyzing biological samples?
A: Yes, HPLC can be used for analyzing biological samples, such as proteins, peptides, nucleic acids, and metabolites. Proper sample preparation and method optimization are essential for successful analysis.